|
This chemical energy supports the light-independent reactions and fuels the assembly of sugar molecules. The real problem is that rotenone prevents the transfer of electrons from NADH to the electron transport chain by. The overall function of light-dependent reactions is to convert solar energy into chemical energy in the form of NADPH and ATP. Additional methods for the identification of plant pigments include various types of chromatography that separate the pigments by their relative affinities to solid and mobile phases. By extracting pigments from leaves and placing these samples into a spectrophotometer, scientists can identify which wavelengths of light an organism can absorb. Spectrophotometers measure transmitted light and compute from it the absorption. An instrument called a spectrophotometer can differentiate which wavelengths of light a substance can absorb. When studying a photosynthetic organism, scientists can determine the types of pigments present by generating absorption spectra. So part of its action is like a molecular motor.\): Plants that commonly grow in the shade have adapted to low levels of light by changing the relative concentrations of their chlorophyll pigments. The current model of its action is called the binding charge mechanism, and it appears that part of this large protein complex accomplishes a mechanical rotation in the process of phosphorylation and release of the ATP molecule. It transports a proton down the gradient and uses the energy to complete the phosphorylation of ADP to ATP. This complex makes use of the proton potential created by the action of the electron transport chain. This protein complex makes use of the metal ions iron and copper in the operation of this electron transfer. 
This makes a total of 10 protons across the membrane for one NADH into the electron transfer chain. This final complex in the electron transport chain accomplishes the final transfer of the electrons to oxygen and pumps two protons across the membrane. More detailĬomplex IV ( Cytochrome c oxidase). Four hydrogens are pumped across the membrane to the intermembrane space. This complex accomplishes the oxidation of ubiquinol and the reduction of two molecules of cytochrome-c. More detailĬomplex III ( Q-cytochrome c oxidoreductase). This complex forms a second entry point into the electron transport chain using the succinate product of the TCA cycle. More detailĬomplex II ( Succinate-Q oxidoreductase). The process accomplishes the pumping of four protons across the inner mitochondrial membrane to the intermembrane space. Electrons are transferred through Complex I using FMN (flavin mononucleotide) and a series of Fe-S clusters. The reduced coenzyme NADH binds to Complex I and accomplishes the reduction of Coenzyme Q10. The emergent picture is that of coupled reactions through five protein structures associated with that inner membrane.Ĭomplex I ( NADH-coenzyme Q oxidoreductase). The electron transport chain is the final stage of aerobic respiration leading to the forming of ATP in the inner membrane of the mitochondrion. Many years of effort have been devoted to the study of the remarkable processes in the mitochondria. The Protein Complexes of the Electron Transport ChainĮlectron transport in the chloroplast for photosynthesis Coupled Systems: The Electron Transport Chain and Oxidative Phosphorylation The protein complex ATP synthase then makes use of this membrane potential to accomplish the phosphorylation of ADP to ATP. This difference in proton concentration produces both an electrical potential and a pH potential across the membranes. This process was called chemiosmosis by its discover, Peter Mitchell. The energy used in the electron transport chain pumps protons across the inner mitochondrial membrane from the inner matrix to the intermembrane space, producing a strong hydrogen concentration gradient. This series of coupled reactions is often referred to as oxidative phosphorylation. These small oxidation steps accomplish the conversion of ADP to the energy currency molecule ATP. 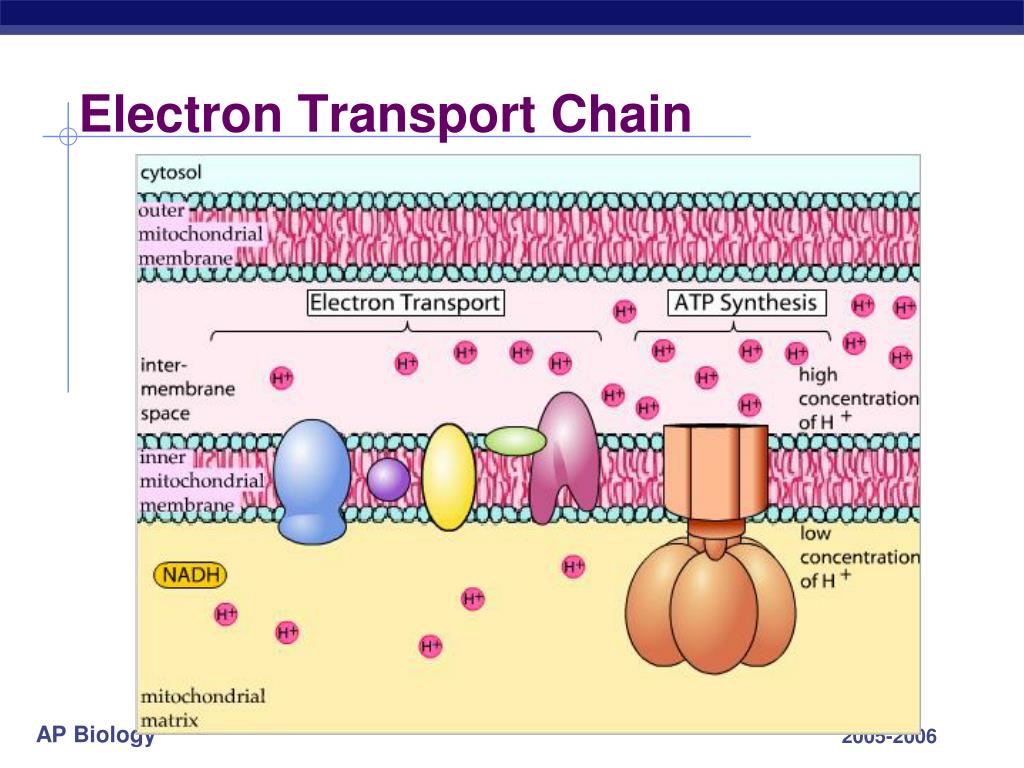
The energy given to the electrons of the reduced coenzyme NADH and to succinate by the TCA cycle is transferred in small steps in the inner membrane of the mitochondrion through a chain of five protein complexes. This cycle deposits energy in the reduced coenzymes which transfer that energy through what is called the electron transport chain. After glycolysis, the pyruvate product is taken into the mitochondia and is further oxidized in the TCA cycle. 
The eukaryotic cell's most efficient path for production of vital ATP is the aerobic respiration that takes place in the mitochondria. Electron Transport in the Energy Cycle of the Cell Electron Transport in the Energy Cycle of the Cell
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
